By Shaoyu Chang, MD, MPH, Senior Research Manager, LSN

Year 2015 has been buzzing with biopharma licensing deals. Compared to previous years, large pharmaceutical companies have become more aggressive in pursuing early stage assets to replenish their pipelines. Against the backdrop of headline-grabbing mega deals, we have dug into the LSN Licensing Deals Platform to examine the key data points that can tell us about the overall deal-making activity for year 2015. (LSN’s data covers licensing deals, but not M&A activity).
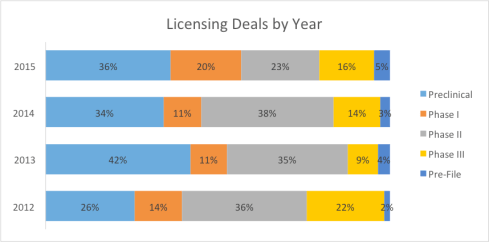
Growing interest in preclinical and Phase I assets
While previously big pharma companies were turning to de-risked projects, this year we have seen a return of interest in early stage assets. Preclinical and phase I projects accounted for over half of the licensing deals. This optimism is partially fueled by scientific advancements such as gene and cell therapies. It is important for startup entrepreneurs to observe the trend and identify strategic partners even at a very early stage.
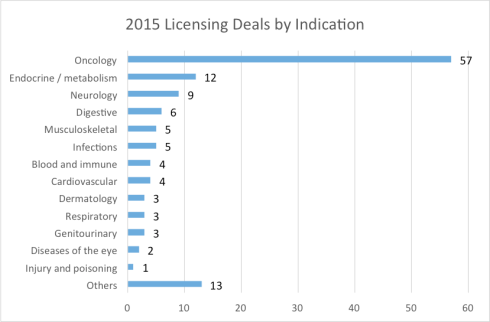
Cancer dominated licensing deals, while diabetes gained ground
Continuing the trend in 2014, cancer continues to take center stage in licensing deals, accounting for two out of five deals in 2015. CAR-T therapies and checkpoint blocking antibodies were some of the most highly sought themes. However it is noteworthy that diabetes (an endocrine disease) has attracted significant interest this year, with a number of mega deals with major multinational giants.
Promising therapeutic breakthroughs attracted big pharma’ interest
The LSN Licensing Deals Platform also contains information about compound type and technology approach that allows the user to dive deeper into in each deal. Across a variety of therapies, a few areas clearly stood out: antibodies, immunotherapy, and gene therapy.
Antibody therapies have been developed to attack novel disease targets such as PD-1 and CD20. Researchers are testing bispecific antibodies and antibody-drug conjugates to achieve higher therapeutic efficacy and specificity in cancer and other diseases.
Cell therapy and immunotherapy have attracted significant interest in 2015, particularly the chimeric antigen receptor (CAR) T cell therapy. With encouraging clinical trial data announced at the American Society of Hematology annual meeting, we expect the trend to continue into 2016.
Recent breakthroughs in gene therapy such as CRISPR has gained traction in year 2015. In addition to cancer, pharmaceutical companies are eager to test these new tools in treating rare diseases such as cystic fibrosis, hemophilia, and sickle cell disease.
Global playing field for big pharmas and entrepreneurs
One would not be surprised to find some of the most active big pharmaceutical companies through the licensing deals LSN tracked. Johnson & Johnson, Celgene, Sanofi, AstraZeneca, and Novartis are among the list of top dealmakers for 2015. Big pharmas are increasingly looking global, for example this year Johnson & Johnson struck deals in the US, Canada, Sweden, and Korea.
Data from the LSN Licensing Deal Platform demonstrated a growing number of emerging market players entering the game in search for partnerships. Hanmi Pharmaceutical is probably the most successful example when it comes to out-licensing. This year the South Korean company has struck deals with four multinational giants totaling more than US$4 billion.
Biotech entrepreneurs are now entering a playing field that is looking optimistic with ample capital and exciting breakthroughs. As we continue to track licensing activities, our best advice for entrepreneurs is to keep partnerships in mind at an early stage of your venture and execute an effective outreach campaign that covers across a broad spectrum of strategic partners.