Diana Saraceni |
An interview with Diana Saraceni, Co-Founder & General Partner, Panakes Partners
– By Karen Deyo, Investor Research Analyst, LSN |
 Karen Deyo |
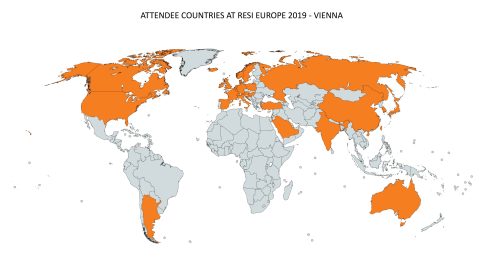
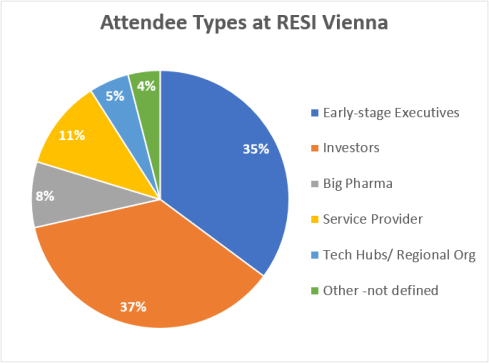
One question that is commonly addressed during RESI panel sessions is: What should a company NOT do in initial meetings with investors? At RESI Europe, Diana Saraceni was a speaker on the Medical Device Investors panel and offered a list of six mistakes companies often make – some all the more important since they are mentioned less frequently. I spoke with Diana following RESI Europe, to recap this important advice.
Karen Deyo (KD): Thank you again for speaking on the panel, Diana. Let us start with the more basic, well-known advice. What are some of the things companies do in discussions with you that send up red flags?
Diana Saraceni (DS): One common issue we see is either too much or too little background information in an introductory pitch deck. On one hand, you do not need to spend several slides discussing what diabetes is, its prevalence and how it is currently treated. On the other hand, make sure to give an overview of the current standard-of-care, as a way to highlight what makes your technology unique. Another issue is a lack of flexibility with regards to valuation. Especially in early-stage, pre-revenue companies, even if they have received advice from a third party, this is never set in stone. You have to be open to negotiate with the investor, and remember that they and their experience are an added value for you.
KD: Any more commonly discussed mistakes you see in companies when approaching investors?
DS: Many companies want investors to sign an NDA before starting any sort of conversation. There is a common misconception that investors will copy your business or steal your idea, but in reality, they are not going to take the time and effort to do so. Signing an NDA before showing unpublished data or starting deep due diligence, both scientific and financial, is accepted, but do not expect an investor to sign an NDA before your first meeting. In addition, don’t expect detailed feedback on your technology or business plan unless the investor has done some sort of due diligence. Remember that investors themselves often turn to experts and advisors when evaluating companies so it is up to you to make sure to consult with KOLs before reaching out to investors.
KD: What are the less commonly discussed, but equally important, errors in fundraising strategy that you’ve seen?
DS: First, when starting your fundraising campaign, don’t immediately go to your top-tier targets right from the beginning. Start with smaller, more flexible, investors who can point out what questions need to be addressed in your initial presentation, helping you hone your pitch. Lastly, if a deal falls through after some due diligence, it is critical to follow up and find out what problem the investors identified. This can be an opportunity to initiate discussions in the future, by coming to the investor with the issue solved. It is important to remember that investors take note of their reasons for passing on an investment opportunity, and they will know if you approach them again without having addressed those issues, and that will not help your case.
KD: Thanks for all of this valuable feedback! I look forward to hearing what other insights you can provide at future RESI events!
 Diana Saraceni is an investor with long experience in Venture Capital. She is currently Co-Founder & General Partner at Panakes Partners (www.panakes.it), an Italian Venture Capital firm dedicated to early stage investments in healthcare in Europe and Israel. Diana can rely on over 15 years of Venture Capital experience. She has also co-founded and managed 360 Capital Partners (www.360capitalpartners.com), one of the leading Venture Capital firm in Europe. She has led investments in a significant number of companies all over Europe, not only in the medical device sector, and generated several exits as IPO and M&A transactions. Ms Saraceni has a strong knowledge of the Venture Capital community in Italy and Europe. Ms. Saraceni holds an Msc in Engineering and an MBA from Luiss University.
Diana Saraceni is an investor with long experience in Venture Capital. She is currently Co-Founder & General Partner at Panakes Partners (www.panakes.it), an Italian Venture Capital firm dedicated to early stage investments in healthcare in Europe and Israel. Diana can rely on over 15 years of Venture Capital experience. She has also co-founded and managed 360 Capital Partners (www.360capitalpartners.com), one of the leading Venture Capital firm in Europe. She has led investments in a significant number of companies all over Europe, not only in the medical device sector, and generated several exits as IPO and M&A transactions. Ms Saraceni has a strong knowledge of the Venture Capital community in Italy and Europe. Ms. Saraceni holds an Msc in Engineering and an MBA from Luiss University.

 The day before RESI Philadelphia, the 2019 Taiwan Biotech Forum will be held at Temple University Science Education and Research Center on June 2nd, 2019.
The day before RESI Philadelphia, the 2019 Taiwan Biotech Forum will be held at Temple University Science Education and Research Center on June 2nd, 2019.