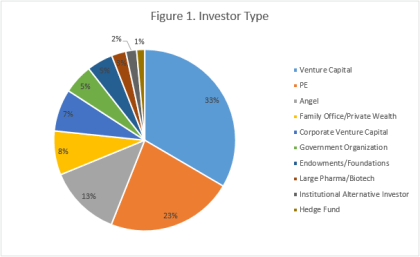
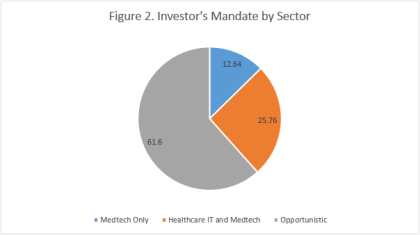
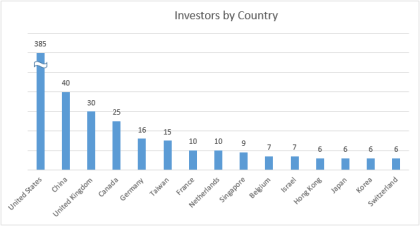
By Dennis Ford, Founder & CEO, LSN

Up to two hundred early stage life science investors will converge on MaRS Discovery District on June 23rd in Toronto. The Redefining Early Stage Investments (RESI) conference series continues to match up fundraising CEOs and scientist-entrepreneurs with global investors who are a fit for their sector and stage of development.
LSN created RESI to fill a hole in the early stage arena by connecting investors and startups at events held every 90 days throughout North America. RESI on MARS is shaping up to be a very compelling event. RESI continues to receive great support from the global early stage life science investor community because investors find great technology assets and startups at RESI, as well as being able to network with other likeminded investors.
Quotes I Heard at RESI Events
Family Offices
An investor representing a large family office who has been to last 5 of our conferences stated he had 16 partnering meetings at a recent RESI event. The one thing he appreciated most was that majority were from outside the traditional biotech hubs – with far more realistic on valuations than those elsewhere.
An investor from a California-based family office who had never been to RESI before said “I go to many conferences across various industries and this was a great combination of useful topics and the kind of opportunities I want to see.”
Corporate VC
A corporate VC investor said he made one investment in a company he met at RESI last year, and this year he found 2-3 he said he was very interested in pulling the trigger on.
A new corporate venture fund from the insurance industry said he needs venues like RESI where he can find next generations solutions for all future applications, and also meet investors and strategic partners for corporate initiatives.
Another corporate VP described RESI as a great venue for finding technology assets. The corporation invested in a company that was sourced from a previous RESI event.
Seed Fund
Funded by Chinese pharma firms looking for early stage investments, this fund’s partner postponed a trip back to China to come to RESI to find regional healthcare technology startups.
Pharma
A U.S. Pharma BD and Licensing lead said “RESI is great for showing what great resources are in second tier regions. I have met several companies at RESI that I will continue to investigate.”
A VP of Outsourced Technology said “I need to find companies before they get tied up with long term VC relationships. RESI enables me to broaden my view of companies in a region I need to canvass. I need to get to know these companies at an early stage.”