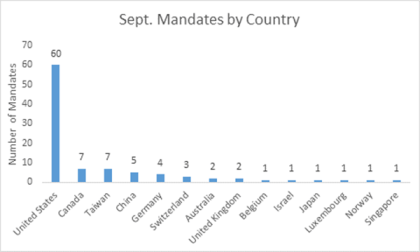
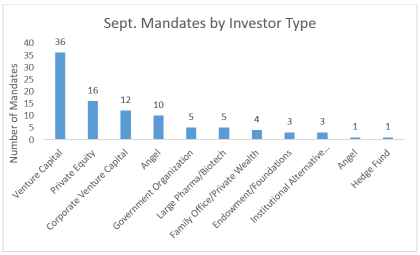
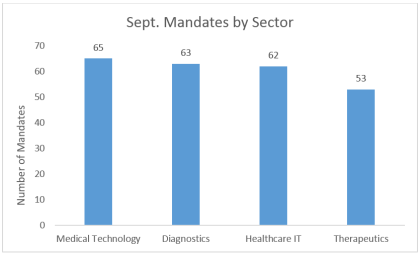
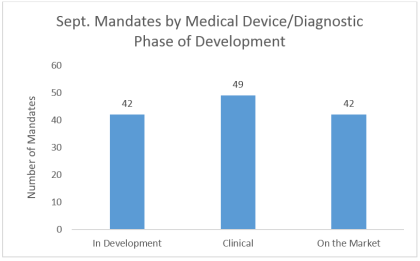
By Shaoyu Chang, MD, MPH, Senior Research Analyst, LSN
An increasing number of innovative entrepreneurs are finding capital, development partners, and distribution channels outside of their geographic region. Who is investing outside of their geographic regions? What are their motivations? What are the benefits and risks when working with cross-border investors? Six experts from prominent VC funds and corporations gathered at the Redefining Early Stage Investments (RESI) conference in Boston on September 16 to address these questions.
Moderated by Lisa Rhoads, Managing Director, Easton Capital, the panel featured:
- Christopher Kim, Managing Partner, Korea-Seoul Life Science Fund
- Luis Barros, Managing Partner, Leading Business Ventures & Venture Partner, Portugal Capital Ventures
- Winston Town, Vice President, Diamond BioFund
- Weiyong Sun, Senior Director, External Scientific Affairs, Daiichi Sankyo Group
- V. Kadir Kadhiresan, Vice President, Venture Investments, Johnson & Johnson Innovation – JJDC
Emerging economies entrepreneurs: aiming for the global market
Many emerging countries have recognized the potential of biotech industries to enhance their own economy. Given the limited size of their domestic markets, startup companies from these countries tend to be valued based on their capability to do business on a global scale.
Several bioclusters have made impressive achievements in terms of scientific discoveries and patent filings. For example, Korea Advanced Institute of Science and Technology (KAIST) is one of the top 10 universities in the world with the highest number of IP. However, bottlenecks exist, especially on the operations side.
The panelists emphasized the three components to build a successful biotech business: innovative science, capital, and the operation team. The panelists recalled some challenges they faced when recruiting the right operation talents for their domestic portfolio companies. Operation, development, and commercialization expertise is sorely needed.
Several models are being employed, from entrepreneurship training sessions to entrepreneurs in residence (EIR). The panelists stressed the importance of cross-border collaborations that can allow businesses to leverage non-dilutive funding from local sources and gain access to strategic markets.
Developed economies entrepreneurs: finding strategic partners
The Asia-Pacific region has attracted attention in recent years due to its sheer market size. A growing number of investors from this region are looking into North America and Europe for advanced technology and strong financial return.
A panelist noted that “[t]he value of a syndication is financial and strategic, and a good co-investor comes in the form of both.” The panelists discussed their experience in co-investing with Asia-Pacific investors.
In one case, Asia-Pacific co-investors were less sensitive to valuation but were focused on specific rights in their domestic market. In another case, Asia-Pacific co-investors were able to utilize their domestic hospital network to aid in clinical development. For early stage ventures focused in developed economies, this created a win-win situation.
Intellectual property is now considered one of the most important piece of value for early stage life science companies. The panelists exchanged their views on intellectual property and addressed the fear of reverse engineering and copyright theft. The panelists emphasized that emerging markets have made significant progress in IP protection over the years. However, partnering with a trustworthy local patent office is still strongly encouraged.
Big pharma: scouting globally, acting locally
The pharmaceutical sector is a global game. Top pharmaceutical corporations in the world are operating and making collaborations on a global scale, whether they are headquartered in the US, Switzerland, or Japan.
Around 15 years ago, strategic financing might account for only 7% of all pharmaceutical deals. Now it hovers at the 35-40% range. As few VCs are willing to take on clinical and regulatory risks, corporate strategics are becoming critical for early stage life sciences.
Big pharma has a plethora of investment vehicles to secure a collaboration, from corporate venture capital funds to joint ventures. When entering a new market, big pharma often partners with local VC to help them acclimate to local culture and oversee the growing business. Entrepreneurs should carefully navigate the changing landscape to choose the right strategic partner.
Tips from Global Investors
How should entrepreneurs go about interacting with global investors? The panelists gave the following tips:
- Good cross-border investors can bring in both strategic and financial value for early stage companies. In-depth understanding of cultural differences and identifying the potential “fit” are key to successful cross-border deals.
- Entrepreneurs should be capital efficient and meticulously manage their assets by focusing on derisking activities along milestones. Early stage cross-border PI collaborations are highly valuable as they open doors to non-dilutive funding.
- Early stage companies should protect their intellectual property in key markets. Local attorneys familiar with regulations and stakeholders in the region can be extremely beneficial in patent filing and litigation.