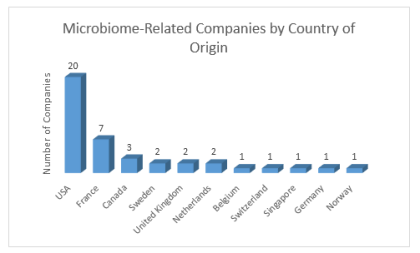
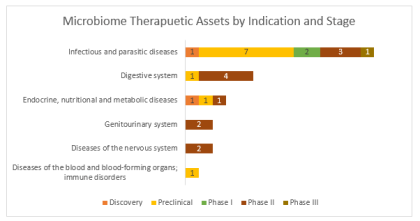
By Lucy Parkinson, Senior Research Manager, LSN
 This week, we are featuring four additional investor mandates, focused on Therapeutics, Medical Device, Diagnostics, and Healthcare IT. LSN Research has seen investors from all across the globe looking for investment opportunities and would like to share more of a sample of the types of information that we gather.
This week, we are featuring four additional investor mandates, focused on Therapeutics, Medical Device, Diagnostics, and Healthcare IT. LSN Research has seen investors from all across the globe looking for investment opportunities and would like to share more of a sample of the types of information that we gather.
1. Therapeutics: US Based Venture Arm of Chinese Family Office Seeking Preclinical Biologic Opportunities
A private investment group that was founded by a single family out of Hong Kong has four operations based in Shanghai, Beijing, Hong Kong and the US. The US-based venture investment arm is actively seeking new investments in the life sciences across the US, and it occasionally invests in companies in Europe. The group focuses on pre-clinical and clinical stage companies. The investment size will vary and typically it ranges from $2 million -$10 million. The group has no set number of allocations over the next 6-9 months.
In the life sciences the firm is looking for new investments primarily in the biotech therapeutics sector. The group will also look at medical devices and diagnostics opportunities, however, which are not their current focuses. For therapeutics, the group is most interested in biologics. The group invests in a variety of therapeutic areas with a focus on oncology. The group generally invests in pre-clinical stage companies without clinical data, and it also syndicates with other investors for clinical-stage investments.
The group invests in privately held life science companies. The group is generally not interested in commercial-stage companies.
If you are interested in more information about this investor and other investors tracked by LSN, please email mandates@lifesciencenation.com
2. Medical Device: Family Office Fund Seeking Early Stage Medical Devices
A family office/venture capital firm based in New York firm manages an evergreen fund that seeks to make seed, venture, and growth equity investments in early to mid-stage medical device companies, but will also consider secured debt to help revenue generating companies with short term cash flow issues. The firm will also consider pre-seed investments. The firm can allocate anywhere from $1M to $10M per company, but generally allocates around $8M. The firm typically does 3 rounds of financing with an initial investment of around $3M. The firm is geographically agnostic, but highly prefers to invest in companies that are based in the US. The firm seeks to make about 3-4 allocations in the next 6-9 months.
The firm is currently looking for medical device companies with a strong emphasis on products with a 510(k) regulatory pathway. In terms of subsectors, the firm is looking for products specifically in the areas of interventional radiology and cardiology, spine/orthopedics and minimally invasive surgery. The firm is generally opportunistic in terms of indication.
The firm is looking to invest in companies with a management team with a track record of success in the healthcare industry. However, the firm is generally flexible and does not require a full management team. The firm can assemble the management team over the course of the investment.
If you are interested in more information about this investor and other investors tracked by LSN, please email mandates@lifesciencenation.com
3. Diagnostics: Global Diagnostics Firm Looking to Make Strategic Investments
A global diagnostics companies is looking to form partnerships and make investments in emerging life science companies. The firm is highly flexible about the structure of its investments, and primarily invests in North America and Europe. The firm looks to act as a long-term, highly activist investor.
The firm primarily invests in the diagnostic imaging sector, including imaging devices and agents, CT and MR technologies, and angiography. The firm prefers to invest in companies at or near the market approval stage but is open to considering investments in earlier-stage companies. In addition to imaging technologies, the firm is also interested in cardiology devices, and other interventional devices including delivery devices.
The firm does not provide passive financing; the firm only invests in companies developing products that the firm would eventually market and sell.
If you are interested in more information about this investor and other investors tracked by LSN, please email mandates@lifesciencenation.com
4. Healthcare IT: VC Arm of Large Conglomerate Seeking HIT and Software Enabled Device Opportunities
The venture and expansion capital arm of a large conglomerate has approximately $270 million in total assets under management. In 2014 the firm closed a fourth venture capital fund at $185 million. The firm is current seeking new opportunities in the life science space. The firm’s initial equity investment range from $3-7 million, but are typically $12-$15 million total through subsequent rounds. The firm will only consider investment into US based firms.
The firm is most interested in the medtech, diagnostics, and information providers spaces. The firm is specifically looking for healthcare IT firms within the information providers space. Medical Devices with a significant software component are also of high interest. The firm is very opportunistic within the areas of diagnostics and medtech, and with that being said the firm would be willing to look at firms that fall within the full gamut of medtech and diagnostics subsectors.
The firm is most interested in early to growth stage companies generally investing in series A and B rounds. For HCIT businesses, the firm typically looks for companies with $3 million or more of revenue. For med tech the firm generally invests in businesses post design freeze and typically does not take product development risk.
If you are interested in more information about this investor and other investors tracked by LSN, please email mandates@lifesciencenation.com