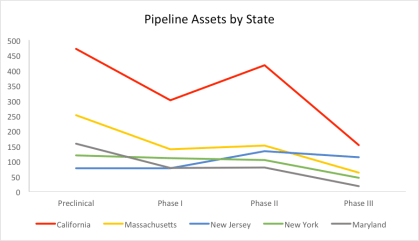
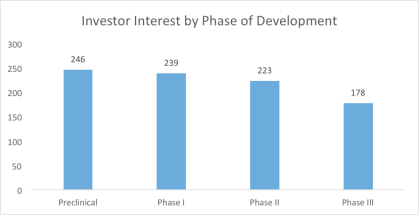
By Jack Fuller, Director of Business Development, LSN
Shaoyu Chang, MD, MPH, Senior Research Analyst, LSN

 LSN and the Texas Medical Center Accelerator have partnered to bring the Redefining Early Stage Investments Conference (RESI) to the emerging life science hub of the American Southwest—Houston, TX—on June 8, 2015. We are proud to announce an open call for applications to the RESI Innovation Challenge. Presenting life science companies will have the chance to showcase their innovations to RESI’s audience at the Texas Medical Center’s new life science accelerator TMCx.
LSN and the Texas Medical Center Accelerator have partnered to bring the Redefining Early Stage Investments Conference (RESI) to the emerging life science hub of the American Southwest—Houston, TX—on June 8, 2015. We are proud to announce an open call for applications to the RESI Innovation Challenge. Presenting life science companies will have the chance to showcase their innovations to RESI’s audience at the Texas Medical Center’s new life science accelerator TMCx.
If you are a scientist-entrepreneur or fundraising CEO starting to accumulate compelling proof-of-concept animal data or early stage clinical data, the RESI Innovation Challenge is for you. The 30 most innovative applicants will be hand selected by Life Science Nation’s internal scientific review board to be part of the RESI Innovation Challenge, presenting their technologies in an exhibition-style format throughout the full-day conference. Presenters will gain additional exposure to investors and potential partners by showcasing their companies and products in a poster format. Unlike traditional 5- to 15-minute pitch presentations that do not allow any real interaction and provide no actionable feedback from investors, the RESI Innovation Challenge enables executives to pitch directly to attendees, generating more in-depth conversations with more investors, more frequently throughout the event.
RESI Innovation Challenge winners are determined by vote of all attendees of the conference. Each attendee receives “RESI Cash” tokens to “invest” in the presenting companies with the most exciting technologies. The company that receives the most investor dollars by the end of the conference is crowned the winner. Previous RESI Innovation Challenge first-prize winners include Empiriko (Newton, MA), ORIG3N (Boston, MA), and Kineta (Seattle, WA).
Houston has emerged as a robust life science hub and gateway to Latin America, and Texas Medical Center is spearheading life science innovation in the region. If you are raising capital in biotech, medical devices, diagnostics, or health IT, you can gain investor exposure by putting your groundbreaking technology under the spotlight at RESI@TMCx.